Editor’s note: Astrobites is a graduate-student-run organization that digests astrophysical literature for undergraduate students. As part of the partnership between the AAS and astrobites, we occasionally repost astrobites content here at AAS Nova. We hope you enjoy this post from astrobites; the original can be viewed at astrobites.org.
Title: Methanimine as a Key Precursor of Imines in the Interstellar Medium: The Case of Propargylimine
Authors: Jacopo Lupi, Cristina Puzzarini, and Vincenzo Barone
First Author’s Institution: Scuola Normale Superiore, Italy
Status: Submitted to ApJ
What Even Is an Imine?
Perhaps one of the biggest questions we can ask is, where does life come from? Many astrochemists seek to answer this question by investigating the history and evolution of molecules that are biologically significant. It turns out imines (pronounced like “I means”) are an important group of molecules that can eventually form DNA. Imines are distinguished by a carbon atom double-bonded to a nitrogen atom, which is then bonded to hydrogen, or “C=NH,” where carbon can be bonded to any other groups of atoms.
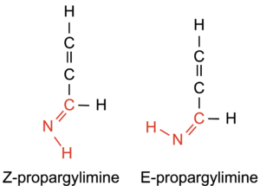
Figure 1: Z- and E-propargylimine. The CNH bonds in red are what classify these molecules as imines. Note that the Z- and E- configurations are different molecules. The slightly different positioning of the N–H bond makes these molecules isomers. [Abygail Waggoner]
Today’s paper uses computational chemistry to determine if the newly detected propargylimine (PGIM) can form via a similar route in the gas phase, or if this large, complex imine is more likely to form in interstellar ices.
Chemistry with Computers
The authors of today’s paper use computational chemistry that uses quantum mechanics to determine the structure and energy of different molecules. The software they used, Gaussian, is commonly used to determine if a chemical reaction is possible and exactly how a set of reactants form a product.
Today’s paper explores many different ways to form PGIM, and they find that the simplest imine, methanimine (CH2NH), is a possible precursor for PGIM. Methanimine can react with either CN or CCH to form CH2NCCH, which then follows a reaction pathway presented in Figure 2 to form either E- or Z-propargylimine. As you can see in Figure 2, these reaction pathways can get pretty complex.
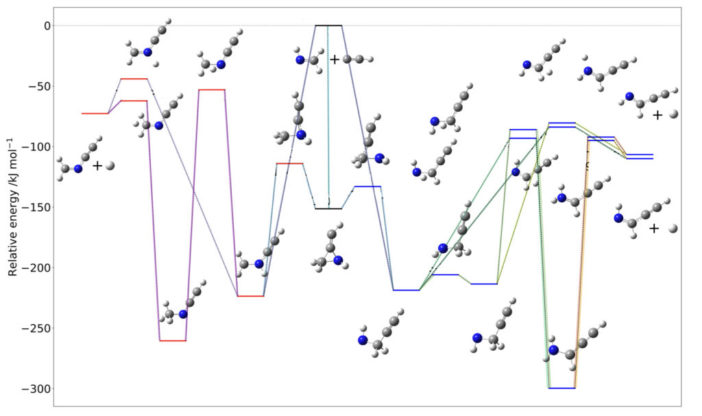
Figure 2: The different reaction pathways to forming PGIM from H2CNCCH and hydrogen. Carbon atoms are represented by black circles, nitrogen by blue, and hydrogen by white. Note that different reaction pathways and branching are represented by different colored lines, and both E- and Z-propargylimine can form. [Lupi et al. 2020]
The type of reaction the authors identified is known as an addition-elimination reaction. Basically, once CH2NCCH is formed from methanimine, a hydrogen atom will be “added” by reacting with CH2NCCH, then the nitrogen and terminal carbon will “switch” spots. Lastly, the hydrogen atom is lost, or “eliminated,” thus forming PGIM.
In addition to the kinematic study shown above, the authors derived the individual rate constants for each step in the reaction pathway shown in Figure 2. The calculated rate constants suggest that the proposed addition-elimination reaction is indeed possible in gas-phase interstellar conditions.
CNH to DNA
So, why is it important that PGIM can form in the gas-phase in the ISM? Well, as can be seen in Figure 2, chemical reactions and reaction pathways are very complex, and many different molecules can be formed many ways. While this study focused on the production of PGIM, the results suggest that other complex imines could form from smaller, less complex imines via a similar pathway in the gas-phase.
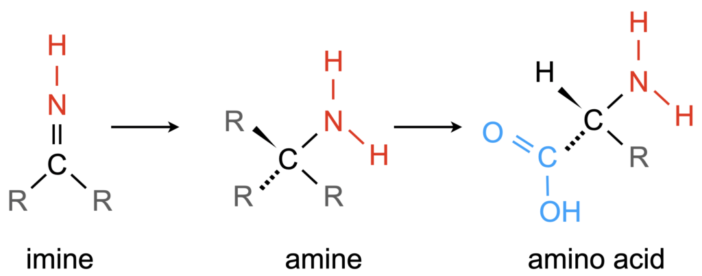
Figure 3: Imines are chemical precursors to amines, classified by carbon bonded to NH2. Amines are chemical precursors to amino acids, which are classified by the NH2 and COOH groups. Amino acids are the building blocks that make up our DNA. In this image “R” indicates any group of atoms. [Abygail Waggoner]
Like we discussed at the beginning of today’s bite, imines are considered a biological precursor to DNA (Figure 3), so it is important to understand their formation to also understand the origins of life in the universe. Traditionally we assume that large carbon-based molecules, like imines, form in the ice. So, the discovery of a possible gas-phase formation route is a new and exciting pathway that could tell us more about the origins of life as we know it.
Original astrobite edited by Huei Sears.
About the author, Abygail Waggoner:
I am a second year chemistry graduate student at the University of Virginia and NSF graduate fellow. I study time variable chemistry in protoplanetary disks. When I’m not nerding out about space, I’m nerding out about fantasy by reading or playing games like dungeons and dragons.
