New laboratory experiments suggest that salty water mixed with Martian surface material can remain a liquid under colder and drier conditions than water alone. This means that liquid water might be found over a larger area of Mars’s surface than previously thought, as well as throughout more of the Martian year, with important implications for habitability and exploration.
The Search for Water on Mars

Warm temperatures on Mars are associated with the appearance of dark streaks on sloping terrain. On Earth, these streaks are caused by water, but on Mars they may be caused by shifting sand grains instead. [NASA/JPL-Caltech/UA/USGS]
But the search continues, with evidence mounting that liquid water might exist in the form of brine: a concentrated mixture of water and salt. Martian brine can form in several ways including by water vapor collecting on the surface of salt crystals. In the lab, researchers have tested the conditions under which brine remains a liquid, rather than freezing or evaporating in Mars’s cold, dry climate. But brine on Mars doesn’t exist in isolation. Instead, it’s muddled together with regolith: the loose mixture of rocks, sand, and dust that coats the planet’s surface. Could the mashup of these two materials help water remain a liquid on Mars’s surface?

An image of Martian soil scooped up by the Phoenix Mars Lander. For this study, the team used simulated Martian soil made from volcanic rocks in the Mojave Desert. [NASA/JPL-Caltech/University of Arizona/Max Planck Institute]
Throwing Regolith into the Mix
To explore this question, Andrew Shumway (University of Washington) and collaborators measured the properties of regolith–brine mixtures in a lab. Since we don’t yet have actual Martian regolith to experiment on, Shumway’s team used a simulated regolith that was originally developed to help NASA scientists test the navigation and sample-collecting skills of the Mars rovers. For their Martian brine, the team swirled together water and a salt called magnesium perchlorate (magnesium and perchlorate are common components of Mars’s surface material).
The team measured two key factors for each of their regolith–brine samples: 1) the freezing point, which partly determines where on the planet’s surface the mixture can remain a liquid, and 2) the amount of water that’s available to participate in chemical reactions and other processes important for life.
Briny Findings
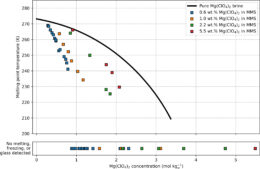
Melting temperature of frozen regolith–brine samples. Samples with a lower melting temperature also freeze at lower temperatures, making them remain liquid under colder conditions. Click to enlarge. [Shumway et al. 2023]
Citation
“Regolith Inhibits Salt and Ice Crystallization in Mg(ClO4)2 Brine, Implying More Persistent and Potentially Habitable Brines on Mars,” Andrew O. Shumway et al 2023 Planet. Sci. J. 4 143. doi:10.3847/PSJ/ace891
